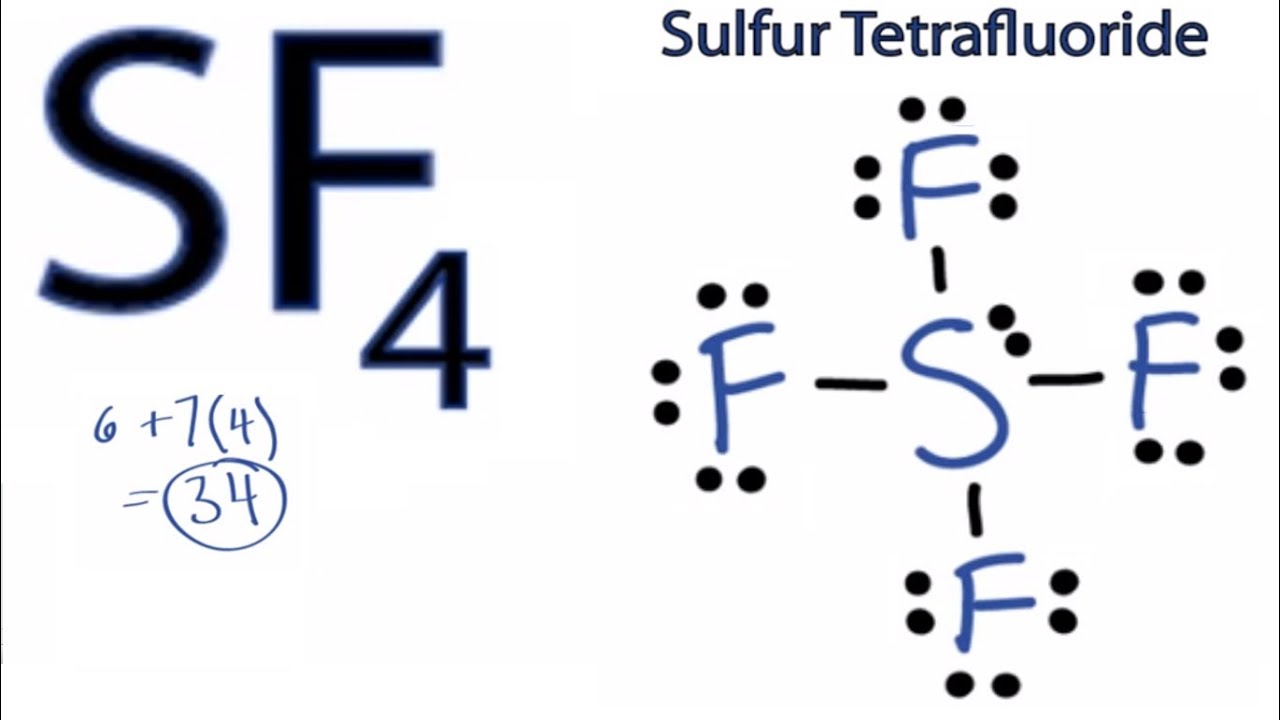
Lithium is a ‘group 1’ element in periodic table with electronic configuration: 1s2 2s1. These two oppositely charged ions form Al2S3 molecule. In this way both of them are able to form stable electronic configuration.Īluminum atom produce positive ion where Sulfur atom become negative ion. These electrons are accepted by Sulfur atom. Six electrons are in 3s and 3p shell of Sulfur.Īccording to Octet rule Aluminum atom transfers these three outer most shell electrons. Lewis dot structure of sulfur tetrafluoride Aluminum and Sulfur lewis dot structureĪluminum is a ‘group 13’ element with outer most shell electronic configuration: 3s2 3p1. The bonding electron pairs between Sulfur and Fluorine are written as straight lines. Sulfur shares its four outer most shell electrons to four electronegative Fluorine atoms. Electronic configuration of Sulfur (valence shell): 3s2 3p4.įluorine atom needs one electron to make stable electronic configuration. It has seven outer most shell electrons in 2s and 2p shell. Sulfur tetrafluoride lewis dot structureįluorine is a ‘group 17’ element in periodic table. The sigma bonds formed between Sulfur and Fluorine written as straight lines. Sulfur donates its six outer most shell electrons to six electronegative Fluorine atoms. So Fluorine atom needs one electron to make it stable like nearest Noble gas. Electronic configuration of Sulfur (valence shell): 3s2 3p4.Īccording to the octet rule every atom tends to fill up the outer most shell with eight electrons. Electronic configuration of Fluorine (valence shell): 2s2 2p5. Sulfur hexafluoride lewis dot structureįluorine is a ‘group 17’ element in periodic table. Each of Chlorine atoms has three lone pairs. Another eight pairs of electron do not take part in bonding. Among these two pairs of electrons form sigma bond. In the molecule three atoms contains total twenty outer most shell electrons. Sulfur electronic configuration: 3s2 3p4. Its electronic configuration (outer most shell): 3s2 3p5. Lewis dot structure of Sulfur monoxide Sulfur dichloride lewis dot structureĬhlorine is a ‘group 17’ element. These electrons are shown by dots around the atomic symbol. For Sulfur there are six valence electrons in 3s and 3p shell. After this we have to count the outer most shell electrons. To draw the electron dot structure of Sulfur atom write the atomic symbol of Sulfur that is “S”. Electronic configuration of Sulfur: 1s2 2s2 2p6 3s2 3p4. Sulfur has sixteen electrons in electronic orbital. From the Lewis dot structure, we can predict different properties of Sulfur. Sulfur is a ‘Group 16’ element in Periodic table. This gives it stable electronic configuration. Sulfur always tends to get two electrons from donor atom. Sulfur is a ‘group 16’ element with six outer most shell electrons. The electrons written as dots may be bonding or unshared electrons. Sulfur lewis dot structure is written by Atomic symbol of that atom. This article is discussing about the Sulfur lewis dot structure involving in bonding with different elements. Lewis dot structure gives information about outermost shell electrons of an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
